LIFE SCIENCES
ENGINEERING SOLUTIONS FOR SUCCESS IN LIFE SCIENCES
When precision and compliance are non-negotiable, you need a partner who understands the critical balance between innovation and regulation. Whether you are engaged in benchtop research, complex manufacturing, or anywhere in between, you require engineered solutions that meet stringent regulatory standards and deliver reliable, repeatable results. With over 35 years of experience in GxP environments, our team provides the expertise, timeline management, and integration services necessary to safeguard product quality, patient safety, and data integrity, allowing you to focus on advancing medical breakthroughs.
Years Serving Life Sciences Clients
Life Sciences Clients
Projects Completed or In-Progress
SPECIALTIES
FDA-Regulated Facilities
cGMP Facilities
GMP Validation Areas + Systems
Environmental Monitoring Systems
21 CFR Part 11 Compliant Process Control Systems
Bio-Safety + Cleanroom R&D Facilities
Hazardous Locations
Combustible Dust Locations
Process + Central Plant Utility Design
Accelerate your medical innovation with streamlined processes.
SOLUTIONS FOR LIFE SCIENCES
Our team of engineers, control system integrators, instrumentation technicians, commissioning and validation experts, and arc flash electrical safety specialists have over three decades of Life Science expertise. With a practical understanding of regulatory standards and a focus on the big picture, we’ll ensure that your operation is not only compliant, but more importantly: safe.
MECHANICAL, ELECTRICAL + PROCESS ENGINEERING
MECHANICAL, ELECTRICAL + PROCESS ENGINEERING
PROCESS CONTROL SYSTEMS
PROCESS CONTROL SYSTEMS
MAIN INSTRUMENT VENDOR
MAIN INSTRUMENT VENDOR
COMMISSIONING, VALIDATION + QUALIFICATION
COMMISSIONING, VALIDATION + QUALIFICATION
MECHANICAL, ELECTRICAL + PROCESS ENGINEERING
We provide precision engineering and project management on every project. Our teams can work alongside other experts for a single system or lead your engineering and process solutions end-to-end, from initial design through operational lifecycle. Our engineering expertise are focused on Good Manufacturing Process (GMP) validation areas, Bio-Safety research settings, cleanrooms, vivarium, and hazardous locations including hazardous dust.

PROCESS CONTROL SYSTEMS
Often called on to solve the most complicated challenges in a facility, Hallam-ICS is known for breaking down the barriers to improvement and success – we can design, build, and program your automation system from start to finish. Our strengths within process controls and plant automation include Batch/S88, facility management, and Plant PAx system.

MAIN INSTRUMENT VENDOR
Our unique combination of engineering, automation, installation, and validation experience provides us with a deep understanding of the needs of our clients from instrument specification through bench testing, installation, field calibration and Turnover Package (ETOP) development.
From procurement and receiving to final delivery, we have the personnel power, cash flow, infrastructure and procedures in place in order to handle a vast project. By engaging Hallam-ICS as a Main Instrument Vendor (MIV) early in the project clients have the advantage of avoiding delays and unnecessary errors in order to stay on track and on budget.

COMMISSIONING, VALIDATION + QUALIFICATION
Our commissioning and validation support Good Manufacturing Practice (GMP) settings, and ensure documented evidence that equipment, software, and instruments used in the manufacture of pharmaceuticals, biologics, and medical devices are fit for use and will provide repeatable functionality.

SUCCESS WITH HALLAM-ICS
Life sciences clients benefit from our engineering expertise, responsiveness, and project management.

CASE STUDY
New Clinical Manufacturing Facility

CASE STUDY
Project Inspire – Durham Clinical Manufacturing Facility
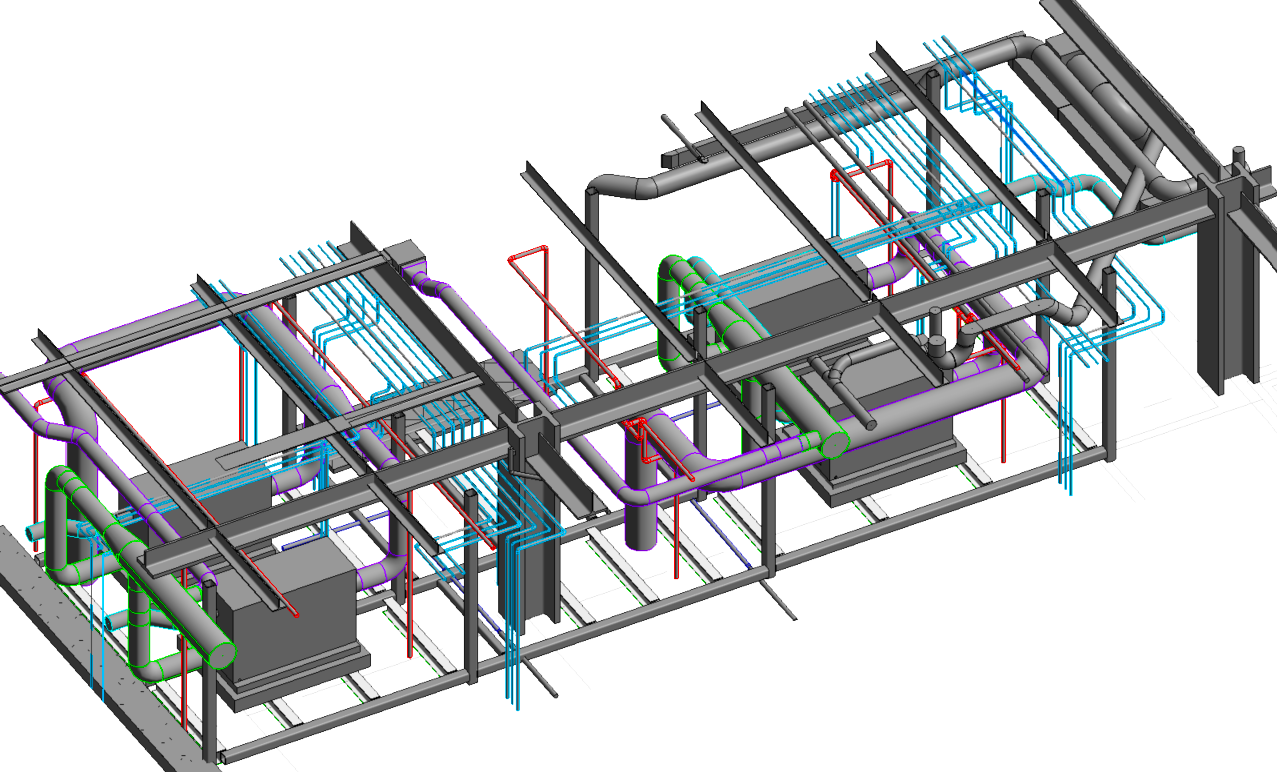
CASE STUDY
Chiller Upgrade Engineering Design
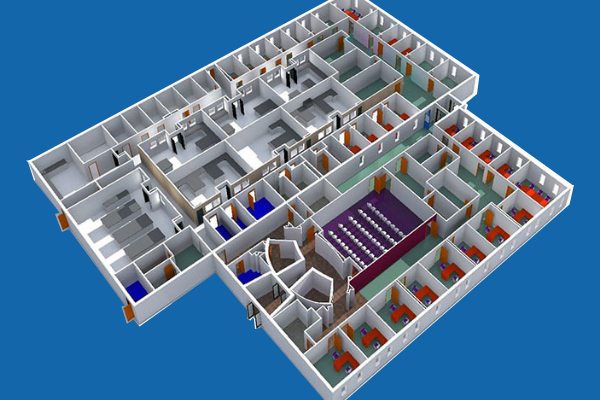
CASE STUDY
New Laboratory Engineering Design

CASE STUDY
B156A Chemistry Labs MEP Engineering Design

CASE STUDY
Siemens Medical Solutions Diagnostics
CASE STUDY
New Manufacturing Facility Instrument Calibration
RELATED RESOURCES
Explore our library of resources to learn more about our exceptional solutions for life science applications.
Frequently Asked Questions
Life sciences engineering and automation solutions are the facility, utility, controls, and compliance-ready systems that enable safe, repeatable operations in FDA-regulated and cGMP environments. Hallam-ICS designs and integrates engineering, process control, and commissioning/validation to safeguard product quality, patient safety, and data integrity.
cGMP facilities commonly require MEP and process engineering for cleanrooms and controlled environments, reliable process/central utilities, and hazard-aware design for regulated and high-risk areas. Hallam-ICS engineers deliver designs that are buildable, maintainable, and ready for qualification.
Commissioning verifies systems operate as intended. Validation and qualification provide documented evidence that equipment, software, utilities, and instruments are fit for use and deliver repeatable performance in GMP environments. Hallam-ICS Cx/V experts plan, document, and execute testing to support compliant startup.
21 CFR Part 11 compliant process control systems support regulated electronic records and signatures (when applicable) through controlled access, traceability, and procedures that protect data integrity. Hallam-ICS designs automation solutions with compliance expectations in mind and aligns documentation for regulated environments.
An EMS monitors environmental conditions that impact product quality—such as temperature, humidity, pressure differentials, and other critical parameters—and documents alarms and trends for investigations and audits. Hallam-ICS integrates EMS platforms with facility utilities and reporting to improve visibility and response.
Batch/S88 is a standard that structures batch control using standardized recipes, phases, and equipment models—making automation easier to validate, maintain, and scale. Hallam-ICS implements batch control strategies and documentation that support reliable GMP production.
PlantPAx is Rockwell Automation’s process control architecture used to standardize PLC/HMI/SCADA development and lifecycle management. Hallam-ICS supports PlantPAx-based solutions for life sciences facilities that want a consistent, supportable platform across multiple lines or sites.
A Main Instrument Vendor (MIV) centralizes instrument specification, procurement, receiving, bench testing, installation coordination, and calibration—so instrumentation doesn’t become the critical path. Hallam-ICS can serve as MIV and develop engineered turnover packages to support qualification and turnover.
An ETOP is the documentation package that supports turnover and validation—typically including device data, calibration records, loop checks, as-builts, and traceability to requirements. Hallam-ICS uses ETOP development to reduce rework and speed up commissioning and qualification.
Hallam-ICS supports FDA-regulated and cGMP facilities, GMP validation areas and systems, biosafety and cleanroom R&D facilities, hazardous locations (including combustible dust), and central utility and process utility systems—bringing engineering, controls, and compliance support under one team.
Hallam-ICS reduces risk by aligning requirements early, building documentation and traceability into design, and planning testing so commissioning evidence supports qualification. Our teams coordinate engineering, automation, and Cx/V to avoid late-stage changes, duplicate testing, and startup delays.
Yes. Hallam-ICS has supported life sciences clients for 35+ years in GxP settings and delivers programs across pharmaceutical, biologic, and medical device operations. We bring practical experience that helps teams move faster without compromising compliance.
If you share your process type, regulatory expectations (GMP/GxP), and project timeline, the Hallam-ICS life sciences team can recommend the right engineering, automation, and Cx/V scope—then provide a clear path to budget and schedule.
OUR TAILORED APPROACH
When the health and safety of individuals is at stake, you need a partner who understands the nuances of your industry. At Hallam-ICS, our tailored approach to the life science industry is based on over 35 years of providing engineering, control systems, and validation solutions that solve the most complex challenges in labs and production facilities. We listen to your goals and come to the table with expertise to engineer a timely solution that safeguards product quality, patient safety, and data integrity. Whether you are engaged in benchtop research or complex manufacturing, our team is equipped to support your facility at every stage, from inception to production.
We follow our proprietary CODES™ process — COMMUNICATIONS, OVERSIGHT, DOCUMENTATION, EXPECTATIONS, and SCHEDULE — to deliver exceptional project quality and consistency. Each project starts with a comprehensive discovery phase, when we gather critical information about your needs and milestones to develop a customized project plan that ensures seamless execution. Throughout the project, we maintain open lines of communication and provide detailed documentation, ensuring that every phase is meticulously managed. Our commitment to technical excellence, combined with our robust customer service, guarantees a successful project outcome and an exceptional customer experience.
FROM OUR BLOG
Stay updated with the latest insights, trends, and expert engineering advice on our blog.

GLP Temperature and Humidity Data Migration During BAS Upgrades: Why It Matters

Autoclave Qualification Explained: IQ, OQ, and PQ for Sterile Compliance



